Does length of salt bridge affect voltage?
The lengths different by a couple cm (12, 9.5, 5, 2). And what I observed was that the smaller the salt bridge, than the higher the voltage.
Does length of salt bridge matter?
Analysis of variance shows that salt bridge length has significant effect both on mean (with 53.90% contribution at 95% CL) and variance (with 56.46% contribution at 87% CL), whereas the effect of cross sectional area of the salt bridge and the interaction of these two factors is significant on mean only (with 95% CL).How does the concentration of the salt bridge affect voltage?
They were connected with a sodium nitrate salt bridge of varying concentrations. As the concentration doubled 5 times from 0.125M to 2M the voltage also increased.How does salt affect voltage?
Because seawater is salty, containing 60 to 100 times more ions than freshwater, it increases the electrical potential, or voltage, between the two electrodes. That makes it possible to reap far more electricity than the amount used to charge the battery.What affects salt bridge?
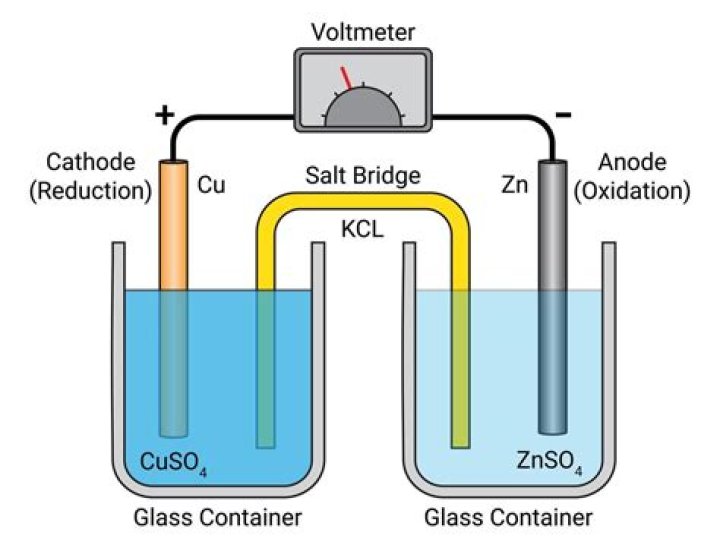
Salt bridges are interactions of amino acids with opposite charge where at least two heavy atoms lie within a hydrogen bonding distance1,2. Often found in solvent exposed parts of proteins, they are susceptible to external interactions, primarily with water.Describe the function of a salt bridge.
What is the length of a salt bridge?
The distance between the residues participating in the salt bridge is also cited as being important. The distance required is less than 4 Å (400 pm). Amino acids greater than this distance apart do not qualify as forming a salt bridge.How does salt bridge affect current?
The current was able to reach the maximum value faster for the smaller salt bridges because less time is required for the sample to distribute into the galvanic cells in smaller bridges than in larger salt bridges. For example, Fig.What factors affect voltage?
The voltage of a cell depends upon a number of factors, including what the electrodes are made from, and the substance used as the electrolyte . A simple cell can be made by connecting two different metals in contact with an electrolyte.Does salt increase voltage?
Adding an electrolyte (like salt) makes the solution more conductive. Greater solution conductivity improves the flow of electricity between the battery electrodes. As a result higher voltage is expected with more electrolyte in the solution.How does distance between electrodes affect voltage?
The results indicate that the distance influences the current density which in turn affects the thicNness of deposition. The higher density of potential lines around the edges gives rise to nonY uniformity in the deposition. The resistance increase due to the higher electrode gap leads to higher energy consumption.Does increasing concentration increase voltage?
In an electrochemical cell, increasing the concentration of reactants will increase the voltage difference, as you have indicated. A higher concentration of reactant allows more reactions in the forward direction so it reacts faster, and the result is observed as a higher voltage.What factors affect the voltage of a galvanic cell?
Factors such as the metals constituting the electrodes play a large role in determining the voltage potential of a galvanic cell, however other variables such as concentration and temperature do affect the voltage produced.Why does voltage decrease when concentration increases?
The voltage decreases when concentration increases because increasing the concentration increases the resistance, decreasing the potential difference.How does surface area of the salt bridge affect voltage in a galvanic cell?
As surface area increases so does the voltage for both cells. As concentration increases so does the voltage for all 3 types of cells. As temperature increases so does the voltage for all 3 types of cells.In which of the following conditions salt bridge is not required in a galvanic cell?
If 2 electrodes are dipped in the same solution, then there is no need for a salt bridge to neutralize the charges because the ions can be easily moved through the porous barriers. We could see the use of salt bridges in galvanic cells such as the Daniel cell or Voltaic cell.Does the salt bridge cause the voltage to be negative?
The electric circuit is completed by the salt bridge, which permits the diffusion of cations toward the cathode and anions toward the anode. Because electrons flow from the tin electrode, it must be electrically negative.How many volts can salt water produce?
Hardly any unbound water molecules are present. The researchers discovered that this saline solution displays an electrochemical stability of up to 2.6 volts –nearly twice as much as other aqueous electrolytes.Does salt increase current?
Dissolving salt in the water creates sodium and chloride ions which in the presence of the potential of the battery provide a path for current flow, the movement of charge. Thus resistance is decreased and current is increased.Is salt a good conductor of electricity?
Conducts ElectricityFor example, solid sodium chloride (NaCl, or table salt) does not conduct electricity; it is an insulator.


