How do you make sodium peroxide?
Sodium peroxide can be prepared on a large scale by the reaction of metallic sodium with oxygen at 130–200 °C, a process that generates sodium oxide, which in a separate stage absorbs oxygen: 4 Na + O2 → 2 Na2O.
Can you make your own peroxide?
Water + air + electricity = hydrogen peroxide.How do you make sodium peroxide from NaCl?
(c) Sodium peroxide First, NaCl is electrolysed to result in the formation of Na metal (Downs process). This sodium metal is then heated on aluminium trays in air (free of CO2) to form its peroxide.What is the use of sodium peroxide?
Sodium Peroxide is a yellowish-white powder. It is used as a bleaching agent, disinfectant, laboratory chemical, and in the production of other chemicals.What's a substitute for peroxide?
The bottom line. Rubbing alcohol and hydrogen peroxide both kill most bacteria, viruses, and fungi. In general, rubbing alcohol is better at killing germs on your hands, as it's gentler on your skin than hydrogen peroxide.Make Sodium peroxide
Can you turn water into hydrogen peroxide?
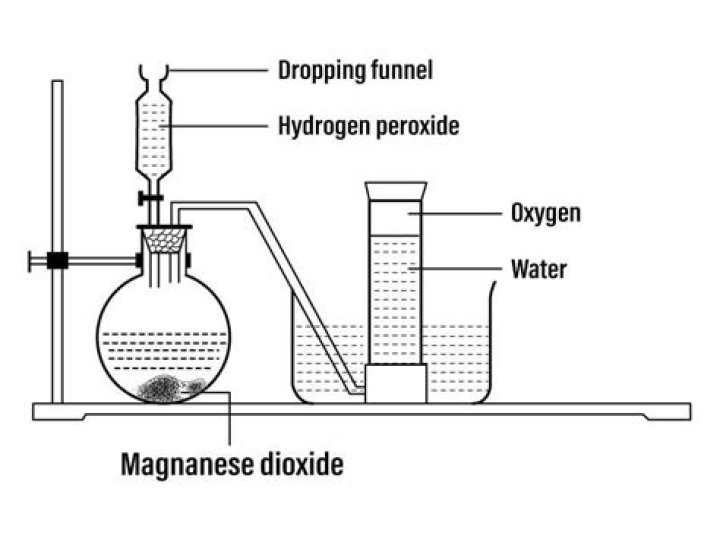
In microdroplets of water, just millionths of a meter wide, a portion of the H2O molecules present can convert into a close chemical cousin, hydrogen peroxide, H2O2, a harsh chemical commonly used as a disinfectant and hair bleaching agent.How do you make hydrogen peroxide solution?
To prepare 100 mL of a 15 mmol/L solution of hydrogen peroxide we will need to dilute 0.15293 mL of 30 % H2O2 to a final volume of 100 mL with deionized (distilled) water. The key concept is that the amount of solute in the desired solution must be equal to the amount of solute in the source solution.What is peroxide made out of?
peroxide, any of a class of chemical compounds in which two oxygen atoms are linked together by a single covalent bond. Several organic and inorganic peroxides are useful as bleaching agents, as initiators of polymerization reactions, and in the preparation of hydrogen peroxide (q.v.) and other oxygen compounds.What are the ingredients of hydrogen peroxide?
Hydrogen peroxide (H2O2) is colorless liquid with a similar chemical formula to water (H2O). The extra oxygen molecule — from which hydrogen peroxide gets its name, as it features one hydrogen per oxygen — represents the main difference between the formulas.Can you mix hydrogen peroxide and lemon juice?
Is It Safe To Mix Hydrogen Peroxide and Lemon Juice? If you're wondering whether or not mixing hydrogen peroxide and lemon juice is safe, worry not! A mixture of both is totally secure and can be used for most purposes that they are used for individuals, but with better effects.Why sodium peroxide is formed?
This yellowish solid is created when sodium is burned in an environment with too much oxygen. It is also a strong base. This metal peroxide exists in many peroxyhydrates and hydrates, including Na2O2·2H2O, Na2O2·2H2O2, Na2O2·2H2O2·4H2O, and Na2O2·8H2O.What happens when sodium peroxide is mixed with water?
"When sodium peroxide dissolves in water, it is hydrolyzed and forms sodium hydroxide and hydrogen peroxide."What happens when sodium peroxide is dissolved in hot water?
Sodium peroxide reacts when it is dissolved in hot water, to produce sodium hydroxide and hydrogen peroxide: 2Na2O2+2H2O(l)→4NaOH+O2(g)How can I make hydrogen at home?
Steps
- Unbend the paperclips and connect one to each terminal of the battery.
- Place the other ends, not touching, into a container of water. ...
- You'll get bubbles off both wires. ...
- Collect the hydrogen gas by inverting a water-filled tube or jar over the wire producing the hydrogen gas.


