Is water a dielectric?
An electrical insulator is a material that does not allow the flow of charge. Charge can flow as electrons or ionic chemical species. By this definition liquid water is not an electrical insulator and hence liquid water is not a dielectric.
Is water a dielectric or conductor?
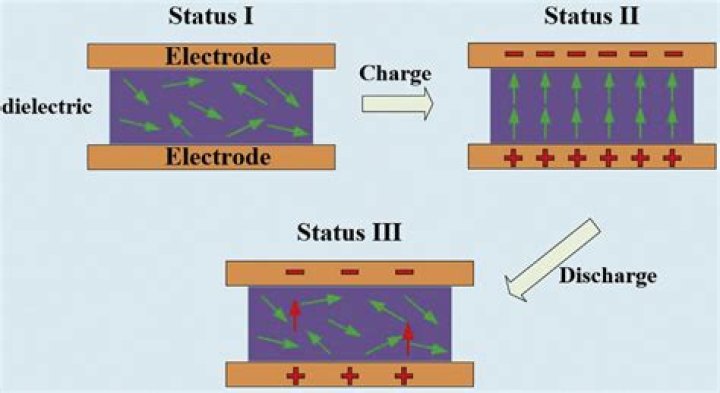
Water comes out to be dielectric because of the dielectric polarization (it's an electric dipole and is a highly polar molecule & even rotates - aligning itself in field direction) associated with it. The electric field induced by polarization overcomes the effect caused by applied electric field.Why is water not used as a dielectric?
Water is not used as a dielectric between the plates of a capacitor, because it has very low dielectric strength and high dielectric constant. It acts as a conductor.Does water have dielectric property?
Answer: Water has a high dielectric constant. Water is unlike any other substance in that it has a very high dielectric constant. The solvent's ability to screen charges is indicated by a high dielectric constant.Is water low dielectric?
Abstract. Liquid water confined within nanometer-sized channels exhibits a surprisingly low dielectric constant along the direction orthogonal to the channel walls.Water as a Dielectric
Why water is a dielectric constant?
Solution : Water has a relatively large dielectric constant because water molecules have permanent dipole moments due to the presence of O-H bonds.Is water have high dielectric constant?
Overview. Water is extremely unique in that it has a high dielectric constant (=80). A high dielectric constant suggests that the solvent (in this case, water) has the ability to screen charges.Is ice a dielectric?
At 0 °C water and ice coexist in a thermodynamic equilibrium. This makes it possible to correctly compare their electric properties and search for the microscopic mechanisms responsible for these properties. Both substances are dielectrics with a wide electronic band gap, ~ 5 eV [1].What are the electrical properties of water?
The Electrical Properties of Water - What You Need to Know
- An Oxygen Atom (8 Electrons + 8 Protons) and a Hydrogen Atom (1 Electron + 1 Proton) ...
- Water: The 8 Electrons in Oxygen's L-Shell are Shared Covalently with 2 Hydrogen Atoms. ...
- The Resistances of Weak Acids and Simple Salts Dissolved in Water.
Is distilled water dielectric?
Abstract. Dielectric constant of distilled water. -The dielectric constant of distilled water was measured with a vacuum tube source of voltage throughout a range of periods extending from about T=1.31×10-8 second to T=8.49×10-8 second corresponding to vacuum wave-lengths of 3.918 meters to 25.47 meters.Is salt water a dielectric?
However, as shown in Fig. 4, the dielectric constant of seawater (aqueous solution of 5 S/m conductivity) is approximately 69, which is less than 90% of that of pure water. In addition, the dielectric constant of water has a strong frequency dependence.Can water be a capacitor?
Applications. A simple type of water capacitor is created by using water filled glass jars and some form of insulating material to cover the ends of the jar. Water capacitors are not widely used in the industrial community due to their large physical size for a given capacitance.Is water electrically conductive?
Water and electricity don't mix, right? Well actually, pure water is an excellent insulator and does not conduct electricity.Does electricity travel through water?
Electricity flows through water because it contains ions of dissolved salts and metals. Distilled water, which does not contain impurities, does not conduct electricity.Can water complete a circuit?
Using tap water, you should be able to complete the circuit and the LED bulb lights up. Using distilled water, however, does not complete the circuit and the LED bulb should not light up.Is air a dielectric?
Some liquids and gases are also good dielectric materials. Dry air is an excellent dielectric and is used in variable capacitors and some types of transmission lines. Nitrogen and helium are good dielectric gases.What is meant by dielectric?
dielectric, insulating material or a very poor conductor of electric current. When dielectrics are placed in an electric field, practically no current flows in them because, unlike metals, they have no loosely bound, or free, electrons that may drift through the material.Which has highest dielectric strength?
A perfect vacuum has the highest dielectric strength, rated at 1×1012 MV/m. A perfect vacuum contains no material to breakdown and is, therefore, the perfect electrical insulator. In reality, a perfect vacuum is nearly impossible to achieve but a high vacuum is also a great insulator, rated at 30 MV/m.What is the dielectric constant of air?
A dielectric constant of 1.00058986±0.00000050 is found for air at normal pressure and temperature.What is permittivity of ice?
The limiting value of permittivity at high frequency is found to be independent of temperature and equal to 3.15±0.05 but there is an increase in the low frequency or static value, from 92 to 103, in polycrystalline ice as the temperature is reduced from 0°C. to −45°C.What is the dielectric constant of ice?
(41-44) Using the experimental value of the dielectric constant of ice at 273 K, i.e., 95, one obtains G = 2.55, in rather good agreement with the value obtained here for the TIP4P models.Why water is a universal solvent?
Because of its polarity and ability to form hydrogen bonds, water makes an excellent solvent, meaning that it can dissolve many different kinds of molecules.Which substance has highest dielectric constant?
Calcium Copper Titanate has the highest dielectric constant.Are water molecules polar or nonpolar?
Water is a Polar Covalent MoleculeThe unequal sharing of electrons between the atoms and the unsymmetrical shape of the molecule means that a water molecule has two poles - a positive charge on the hydrogen pole (side) and a negative charge on the oxygen pole (side).


