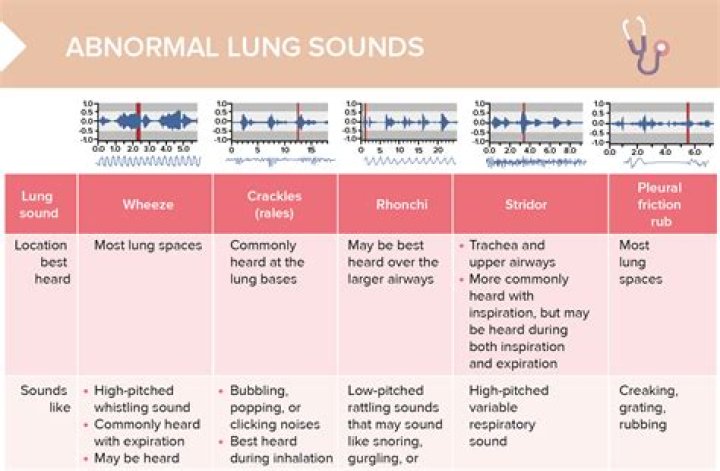
What acid is stronger than muriatic acid?
Hydrochloric acid is a good example for a strong acid. The main difference between hydrochloric acid and Muriatic acid is that hydrochloric acid is composed of only HCl molecules whereas Muriatic acid is composed of impurities along with HCl molecules.
Which acid is stronger muriatic or sulfuric?
The more negative the pKa, the stronger the acid. An acid like sulfuric acid that can give away two hydrogen ions has two pKas. The pKa1 for sulfuric acid is -3, while its pKa2 is 1.99. The pKa for muriatic acid, by contrast, is -7.Is nitric acid stronger than muriatic acid?
Yes, hydrochloric acid is stronger than nitric acid. This is due to the deprotonation of hydrochloric acid in aqueous form. This makes hydrochloric acid stronger than nitric acid.Which acid is the strongest acid?
That makes sulfuric acid the strongest 'normal' acid you'll find. Anything more powerful is defined as a superacid. Superacids are made by taking an already strong (usually Lewis) acid and adding an (often Brønsted–Lowry) acid, which frequently contains the super reactive element fluorine.Is hydrochloric acid stronger than muriatic acid?
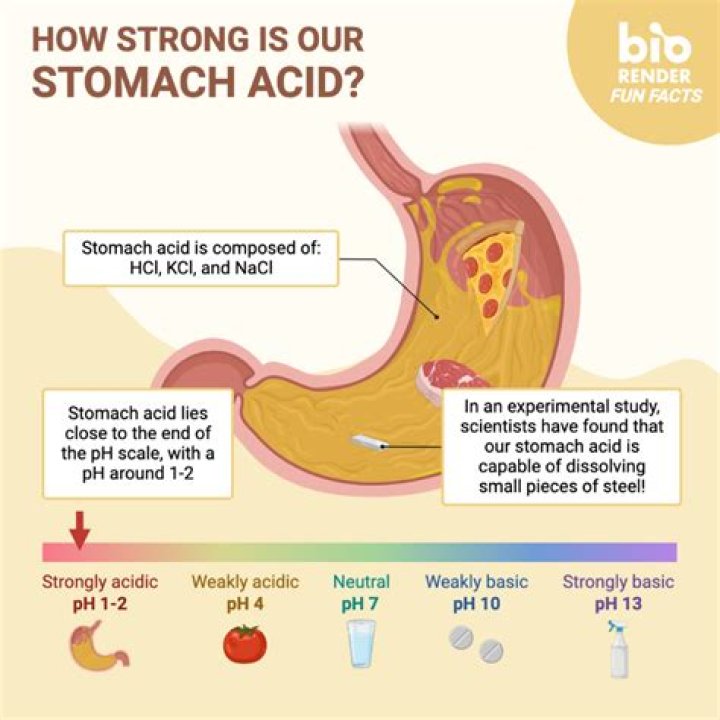
Though still highly corrosive, muriatic acid is milder than hydrochloric acid and is therefore more suitable for home use. At a pH of 1 to 2, muriatic acid can be as acidic as stomach acid (which is also in part comprised of hydrochloric acid) or lemon juice.Top Strongest Acids Ever
What happens if you mix bleach and muriatic acid?
If you mix bleach, or sodium hypochlorite with liberate acid, it will cause chlorine gas, which is toxic, and a severe irritant to eyes and nasal passages, even in small amounts. ... If mixed with chlorine, the acid is deadly.Which acid is king of acid?
Sulfuric acid is commonly supplied at concentrations of 78, 93, or 98 percent. Sulfuric acid is sometimes referred to as the “king of chemicals” because it is produced worldwide in...What are the 7 strong acids?
Here is a glance at all the 7 strong acids and their chemical formulae:
- Chloric acid: HClO3.
- Hydrobromic acid: HBr.
- Hydrochloric acid: HCl.
- Hydroiodic acid: HI.
- Nitric acid: HNO3.
- Perchloric acid: HClO4.
- Sulfuric acid: H2SO4.
What are the 6 strong acids?
- Hydrochloric Acid (HCl) Hydrochloric acid, also known as muriatic acid, is a chemical compound with the formula HCl. ...
- Hydrobromic Acid (HBr) ...
- Hydroiodic Acid (HI) ...
- Sulphuric Acid (H2SO4) ...
- Nitric Acid (HNO3) ...
- Perchloric Acid (HClO4) ...
- Chloric Acid (HClO3)
What are the strongest acids in order?
There are 7 strong acids: chloric acid, hydrobromic acid, hydrochloric acid, hydroiodic acid, nitric acid, perchloric acid, and sulfuric acid. Being part of the list of strong acids doesn't give any indication of how dangerous or damaging an acid is though.What are the 7 strong bases?
The strong bases are:
- LiOH - Lithium Hydroxide.
- NaOH - Sodium Hydroxide.
- KOH - Potassium Hydroxide.
- RbOH - Rubidium Hydroxide.
- CsOH - Cesium Hydroxide.
- Ca(OH) 2 - Calcium Hydroxide.
- Sr(OH) 2 - Strontium Hydroxide.